5 Which of the Following Accurately Describes the Ph Scale
The lower the concentration of hydrogen ions the higher the acidity is. A it measures the acidity of a solution.
C it shows neutral at a pH of 7.

. For each pH unit increase there is a 10-fold decrease in hydrogen ions. Which of the following accurately describes the differing effects the new industrialism had on white-collar and blue-collar workers. Which of the following is NOT correct about the pH scale of a solution.
This accurately describes the pH scale. This value is considered neutralneither acidic or basic. Which of the following accurately describes the pH scale.
The greater the concentration of hydroxide ions the higher the acidity is. The lower the ph is the more basic the solution is. A state of matter that has a definite volume but not a definite shape.
Scientists can accurately predict when an earthquake will occur but not where. The pH of blood ranges from 735-745 while the pH of other bodily fluids varies. The greater the pH is the lower the acidity is.
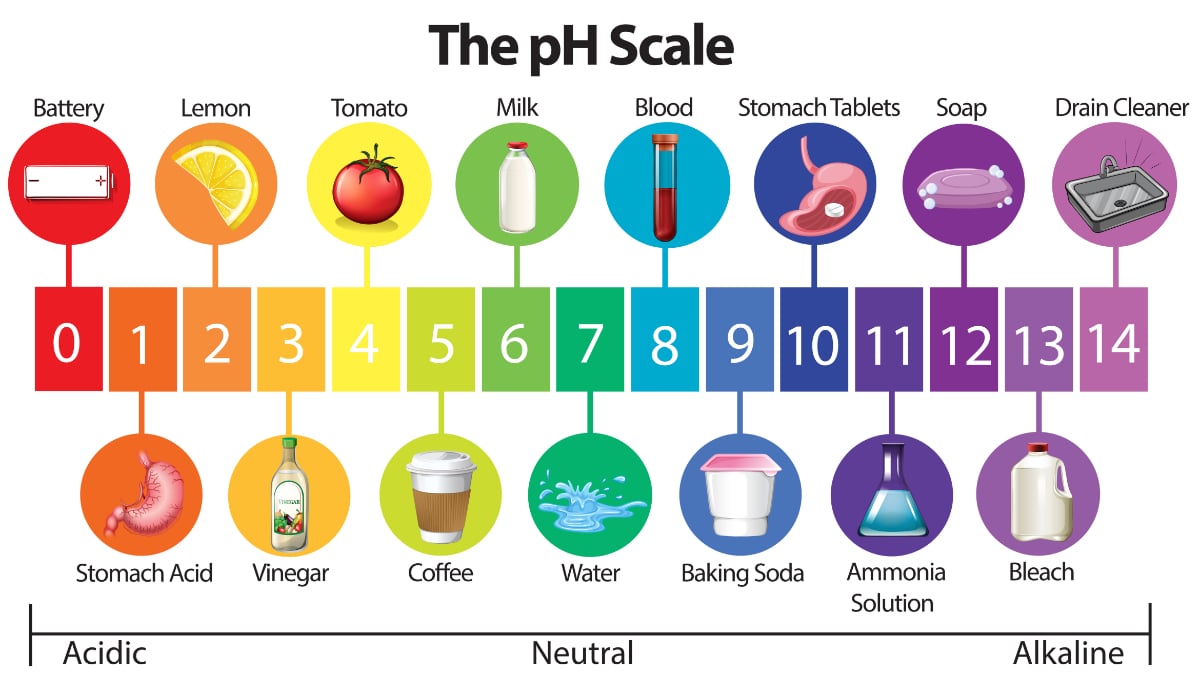
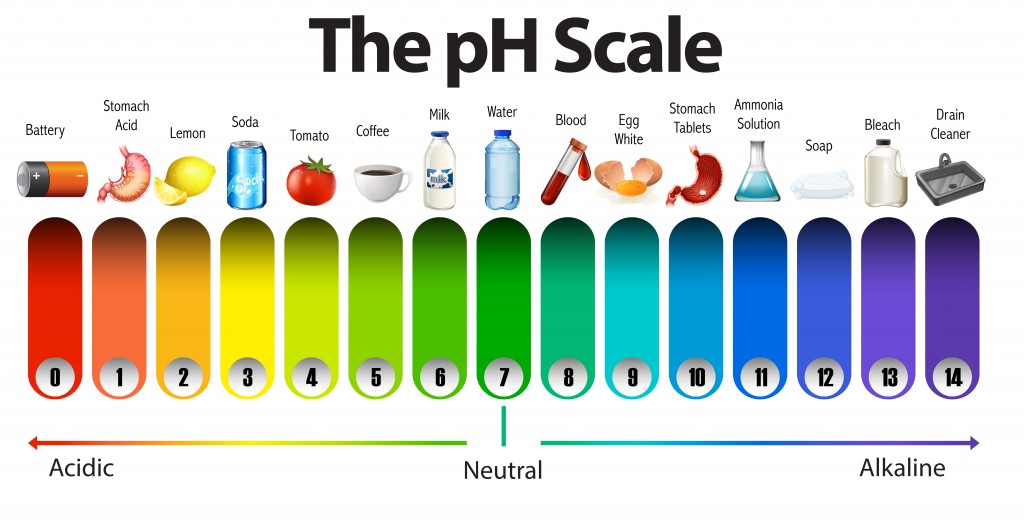
PH scale ranging from 0 very acidic to 14 very basicalkaline and listing the pH values of common substances. A solution with a pH of 4 has twice the H of a solution with a pH of 2. The lower the pH is the more basic the solution is.
Other questions on the subject. Which of the following statements best describes the state of earthquake prediction. Most parts of our body excluding things like stomach acid measure around 72 and 76 on the pH scale a 7 is neutral on the scale.
The lower the concentration of hydrogen ions the higher the acidity is. Scientists can accurately predict the time and location of about 50 of all earthquakes. It could decrease from 70 to 40.
For example pH 4 is ten times more acidic than pH 5 and 100 times 10 times 10 more acidic than pH 6. For each pH unit decrease there is a 10-fold decrease in hydrogen ions. Anything below 70 is acidic and anything above 70 is alkaline or basic.
The pH scale is often said to range from 0 to 14 and most solutions do fall within this range although its possible to get a pH below 0 or above 14. The greater the ph is the lower the acidity is. PH 0 has the highest hydrogen ion H concentration.
Which of the following statements about the pH scale is not true. Correct answer to the question Which of the following accurately best describes the ph scale. The greater the concentration of hydroxide ions the higher the acidity is.
Objects that are not very acidic are called basic. PH 14 has the highest hydroxide ion OH- concentration. The greater the pH is the lower the acidity is.
A state of matter that has free electrons. If foreign strong substances dramatically change this pH our bodies can no longer function properly. Which of the following accurately describes the ph scale.
Which of the following accurately best describes the ph scale. Which of the following accurately describes the pH scale. Which of the following accurately describes the pH scale.
The pH scale is based on a. September 26 2021 thanh. The greater the pH is the lower the acidity is.
The lower the pH is the more basic the solution is. It could decrease from 100 to 70. Coma can occur if the pH falls below 69.
It could increase from 70 to 100. A pH of 7 is neutral. The lower the concentration of hydrogen ions the higher the acidity is.
The greater the concentration of hydroxide ions the higher the acidity is. Investigate how adding more of a liquid or diluting with water affects pH. A pH less than 7 is acidic.
A solution with a pH of 4 has twice the H of a solution with a pH of 2. The greater the concentration of hydroxide ions the higher the acidity is. A state of matter that has both a.
D it indicates basic when the values is greater than 7. As you can see from the pH scale above pure water has a pH value of 7. A pH greater than 7 is basic.
The lower the ph is the more basic the solution is. BBoth types of workers were in low demand. In a pH expression the hydronium ions H3O can be abbreviated simply as H.
The greater the pH is the lower the acidity is. The lower the concentration of hydrogen ions the higher the acidity is. B it has values that the range from 0 to 12.
Scientists can accurately predict the time and location of almost all earthquakes. Both types of workers were in high demand throughout the second industrial revolution. The lower the pH is the more basic the solution is.
The pH scale which measures from 0 to 14 provides an indication of just how acidic or basic a substance is. Which of the following accurately describes the ph scale. Normal clean rain has a pH value of between 50 and 55 which is slightly acidic.
The lower the pH is the more basic the solution is. In a pH expression the hydronium ions H3O can be abbreviated simply as H. Which of the following describes how the pH of the solution could change.
Test the pH of everyday liquids such as coffee spit and soap to determine whether each is acidic basic or neutral. The pH scale ranges from 0 to 14. View more similar questions or.
The pH scale ranges from 0 to 14. The greater the concentration of hydroxide ions the higher the acidity is. Representing Numbers in Base Ten.
The greater the pH is the lower the acidity is. It could increase from 40 to 70. The greater the ph is the lower the acidity is.
The lower the pH is the more basic the solution is. Which of the following accurately describes the pH scale. AWhile skilled workers saw opportunities decline white-collar work expanded.
A state of matter that conducts electricity well. The greater the concentration of hydroxide ions the higher the acidity is. The pH of various bodily fluids on the other hand differsThe pH of saliva is between 65 and 75.
The scale has values ranging from zero the most acidic to 14 the most basic. Low pH suggests that there are too many H ions present while high pH indicates that there are too many OH- ions present. Which of the following is incorrect regarding the pH scale.
The lower the concentration of hydrogen ions the higher the acidity is. The greater the concentration of hydroxide ions the higher the acidity is. The lower the concentration of hydrogen ions the higher the acidity is.
The pH scale is logarithmic and as a result each whole pH value below 7 is ten times more acidic than the next higher value. The greater the pH is the lower the acidity is. Which of the following statements about the pH scale is not true.
The lower the concentration of hydrogen ions the higher the acidity is.

Acids And Bases Ck 12 Foundation
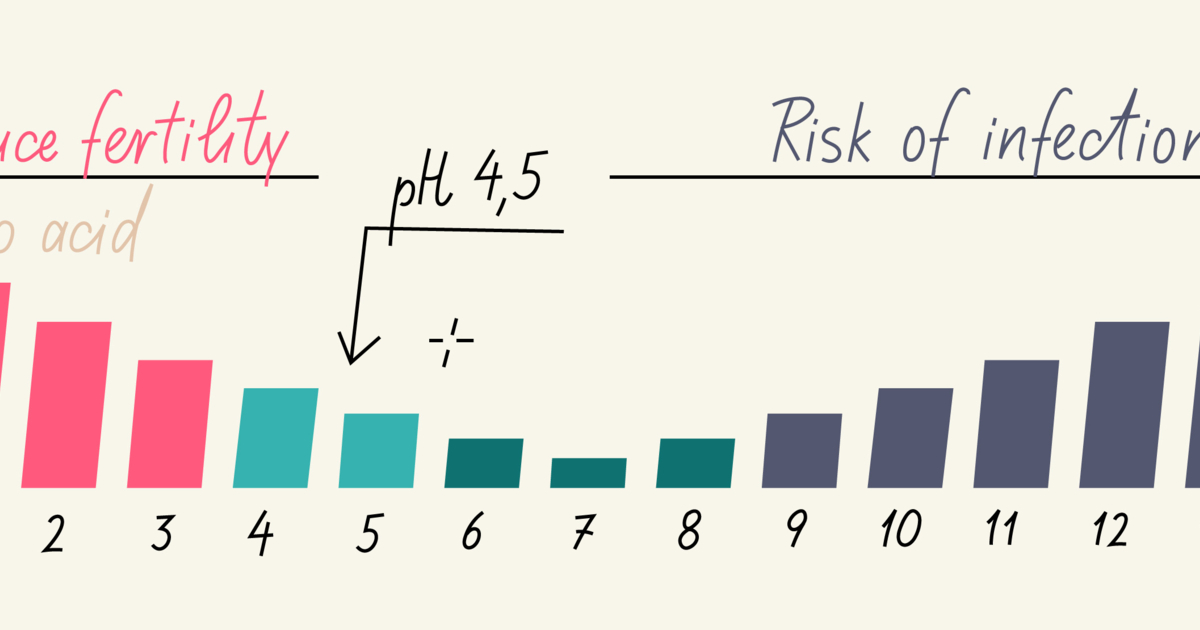
Normal Vaginal Ph How To Test Balance And Restore Vaginal Ph
Examples Of Corrosive Substances And Their Ph Levels

Ph Scale U S Geological Survey

The Acid Base Properties Of Water Video Lesson Transcript Study Com

Soil Ph Level Explained Bbc Gardeners World Magazine

What Is Acidity In Wine Wine Enthusiast

The Four Things You Need To Know About Soil Ph Finegardening Soil Ph Fine Gardening Soil
Water Quality 101 What Is Ph In Water Testing

Renal Function Acid Base Balance Acid Base Disorders Mcq

Acid Base Balance Anatomy And Physiology

The Ph Scale Biology For Non Majors I
Why Does The Ph Scale Range From 0 To 14 Can It Go Beyond That Range
:max_bytes(150000):strip_icc()/pHscaleimageblueringmediagetty-f6fca462fa5f42ec950954275d4ef789.jpg)
9 Natural Remedies To Restore Ph Balance
/LinearRelationshipDefinition2-a62b18ef1633418da1127aa7608b87a2.png)
Linear Relationship Definition

Ph Scale In Simple Terms Youtube

Visual Analogue Scale Vas For Assessment Of Children S Pain Perception Download Scientific Diagram

Pin By Roshan On Ph Scale Remove Toxins Healthy Water Drinks Banner Printing

Normal Vaginal Ph How To Test Balance And Restore Vaginal Ph

Comments
Post a Comment